Non-Invasive Brain Stimulation
Transcranial magnetic stimulation (TMS)
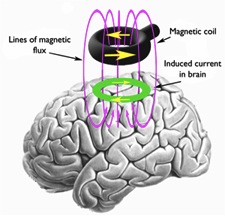 TMS is a noninvasive focal method for modulating cortical activity. It utilizes the principle of electromagnetic induction and involves the discharge of a very large current from a bank of capacitors that rapidly flows through a simple circuit and then a copper-wire coil. This current induces a rapid time-varying magnetic field in the coil. When this coil is held to the head of a subject, the magnetic field penetrates the scalp and skull and induces a small current parallel to the plane of the coil in the brain. When the induced current is sufficient, neuronal membranes depolarize and action potentials are generated. This ability of TMS to modulate cortical activity has made a wide array of applications possible, and a number of studies have demonstrated that TMS can alter functional organization of the human cortex, leading to changes in both the neurophysiologic properties of those brain areas and in the performance of tasks that rely on those regions. There are risks associated with receiving TMS. The most serious of these is the potential to induce a seizure, even in subjects who have no prior predisposition to seizures. Although the risk of seizure induction or other adverse side effects are low, appropriate safety guidelines and precautions must be followed, and individuals administering TMS must be properly trained. Subjects considering participation in a TMS study should know that there are medical conditions that may preclude them from receiving TMS. It is important to discuss these with investigators and to undergo proper screening prior to receiving TMS. View our screening questionnaire.
TMS is a noninvasive focal method for modulating cortical activity. It utilizes the principle of electromagnetic induction and involves the discharge of a very large current from a bank of capacitors that rapidly flows through a simple circuit and then a copper-wire coil. This current induces a rapid time-varying magnetic field in the coil. When this coil is held to the head of a subject, the magnetic field penetrates the scalp and skull and induces a small current parallel to the plane of the coil in the brain. When the induced current is sufficient, neuronal membranes depolarize and action potentials are generated. This ability of TMS to modulate cortical activity has made a wide array of applications possible, and a number of studies have demonstrated that TMS can alter functional organization of the human cortex, leading to changes in both the neurophysiologic properties of those brain areas and in the performance of tasks that rely on those regions. There are risks associated with receiving TMS. The most serious of these is the potential to induce a seizure, even in subjects who have no prior predisposition to seizures. Although the risk of seizure induction or other adverse side effects are low, appropriate safety guidelines and precautions must be followed, and individuals administering TMS must be properly trained. Subjects considering participation in a TMS study should know that there are medical conditions that may preclude them from receiving TMS. It is important to discuss these with investigators and to undergo proper screening prior to receiving TMS. View our screening questionnaire.
Transcranial direct current stimulation (tDCS)
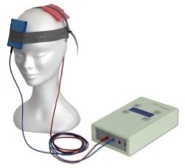 Transcranial direct current stimulation (tDCS) employs a small direct current that is delivered to the scalp, penetrating the skin, skull and meninges to stimulate the brain. These currents have been shown to modify neural transmembrane electrical potentials, leading to transient alteration of neuronal excitability and firing rates. Depending on the use of anodal or cathodal stimulation, firing rates of underlying cortical neurons are either increased or decreased, leading to transient demonstrable changes in perception or behavior. The advantages of tDCS are manifold, and include tolerability, ease of use, portability, and low risk of adverse events, including seizures. Given its advantages, tDCS has been gaining popularity both as a tool for neuroscience research and as a potential modality for the assessment and treatment of a variety of conditions, including neuropsychiatric disorders, Parkinsonism, stroke, and chronic neuropathic pain.
Transcranial direct current stimulation (tDCS) employs a small direct current that is delivered to the scalp, penetrating the skin, skull and meninges to stimulate the brain. These currents have been shown to modify neural transmembrane electrical potentials, leading to transient alteration of neuronal excitability and firing rates. Depending on the use of anodal or cathodal stimulation, firing rates of underlying cortical neurons are either increased or decreased, leading to transient demonstrable changes in perception or behavior. The advantages of tDCS are manifold, and include tolerability, ease of use, portability, and low risk of adverse events, including seizures. Given its advantages, tDCS has been gaining popularity both as a tool for neuroscience research and as a potential modality for the assessment and treatment of a variety of conditions, including neuropsychiatric disorders, Parkinsonism, stroke, and chronic neuropathic pain.



