Amaravadi Lab
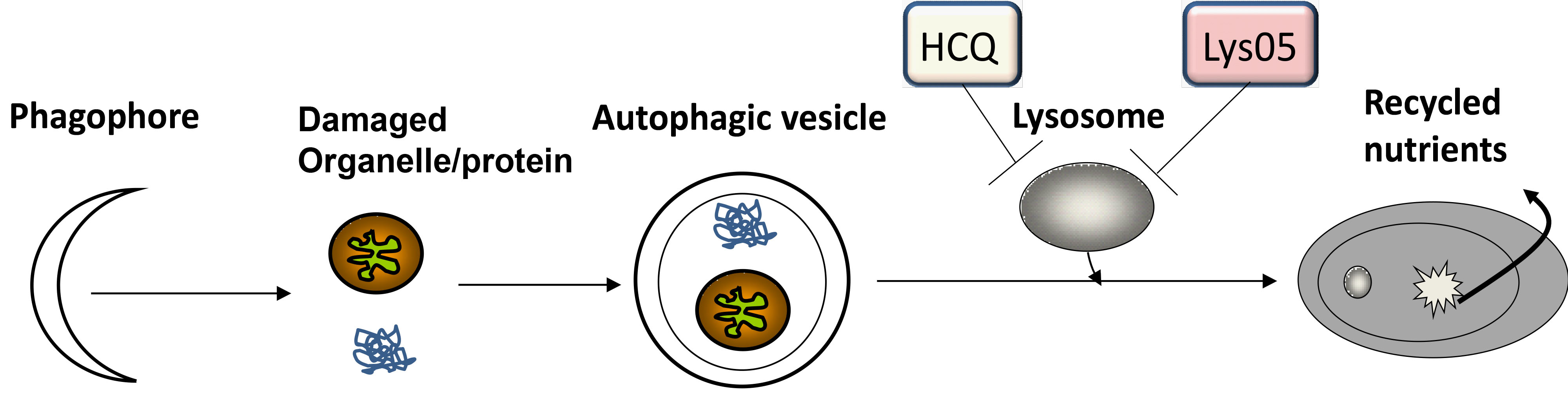
The Amaravadi Lab focuses on developing new cancer therapy that addresses therapy resistance. Cancer therapy has made major strides in the past 20 years, but eventually most cancers have the ability to survive the stress of cancer therapy and recur, leading to suffering and death. We and others have demonstrated that autophagy, the process by which organelles such as mitochondria and proteins are internally digested and recycled, promotes cancer cell survival in advanced cancers, and is a potentially key resistance mechanism that cancers use to survive cancer therapy. Our lab collaborates closely with other basic scientists, clinical researchers, biotechnology and pharmaceutical companies so that we can test our hypotheses in cell lines, 3D culture, mouse models, and patients. We conduct basic laboratory research to understand fundamental aspects of autophagy in cancer biology, but we also have a translational focus. We are actively conducting innovative clinical trials intended to modulate autophagy to enhance effectiveness of existing therapies. Our funding comes largely from the National Cancer Institute, focused pharmaceutical collaborations, and private donations from patients and fundraising programs.
Past Projects
Can more potent and specific lysosomal autophagy inhibitors be identified?
Working with the laboratory of Dr. Jeffrey Winkler (Merriam Professor of Organic Chemistry Department of Chemistry, School of the Arts and Sciences University of Pennsylvania) we have designed, synthesized and tested the first series of dimeric chloroquine derivatives. We call these compounds Lys01 derivatives (see Mcafee et al PNAS 2012). We have identified dozens of potent derivatives (Rebecca et al. Cancer Discovery 2017; Rebecca et al. Cancer Discovery 2019) and identified the molecular target of chloroquine derivatives as palmitoyl-protein thioesterase 1 (PPT1). PPT1 inhibitors are being developed for clinical trials by Pinpoint Therapeutics, Inc.
2041What are the effects of lysosomal autophagy inhibition in specific cell types within the tumor microenvironment?
We recently showed that PPT1 inhibition in tumor associated macrophages enables T cell mediated killing of melanoma tumors (Sharma et al. JCI Insight 2020). Numerous groups have demonstrated that autophagy inhibition at other nodes in the pathway augments anti-tumor immunity. More work is needed to understand how best to use autophagy inhibitors in the context of immunotherapy.
What is the role of PPT1 in tumorigenesis?
Using CRISPR technology we have created novel tools to study the role of PPT1 in tumorigenesis . CRISPR/Cas9 technology was used to insert LoxP sites around exon 1 of the mouse Ppt1 gene to generate conditional Ppt1 knockout mice. Then the mice were crossed into a tamoxifen inducible model of melanoma tumorigenesis. In vivo loss of PPT1 had no effect on initiation or growth of these tumors. CRISPR/Cas9 was also used to knockdown expression of PPT1 in human and mouse melanoma cell lines both constitutively and acutely (Dox inducible). While chemical inhibition of PPT1 with DC661 is toxic to melanoma cells, genetic inhibition was not. Comparison of cells with knockdown by CRISPR/Cas9, siRNA or chemical inhibition indicated distinct proteome changes to adapt to loss of PPT1 which has implications for therapeutic use of PPT1 inhibitors. (Crissey, et. al. Autophagy 2025)
Can we find the cancers and patients most likely to benefit from lysosomal autophagy inhibition?
Using -omics platforms we are investigating, genetic and epigenetic determinants of sensitivity to chloroquine derivatives in a broad panel of tumor cell lines and patient tissues. This work is in collaboration with investigators from the Abramson Family Cancer Research Institute and the Center for Clinical Epidemiology and Biostatistics. Preliminary findings were published (Piao Autophagy 2017), but we continue to investigate candidate markers of sensitivity in human tumor samples.
Can proteins that are secreted in an autophagy dependent manner be used to develop biomarkers of response to autophagy inhibitors?
Working with laboratory of Dr. David Speicher (Caspar Wistar Professor of Computational and Systems Biology, Wistar Institute), we have probed the secretome of melanoma cells grown in 3D culture and have identified autophagy dependent secreted proteins (Kraya et al Autophagy 2015). We are currently determining effects of therapeutics that are known to modulate autophagy on secretome profiles. We are working on both small molecule targeted therapeutics and immunotherapies in immunocompetent mouse models.
How exactly does targeted therapy activate a cytoprotective autophagy response?
Working with the laboratory of Dr. Costas Koumenis (Professor and Vice Chair for Research Department of Radiation Oncology, University of Pennsylvania) we previously determined that BRAF inhibitors activate cytoprotective autophagy through an ER stress response. ( Ma et al JCI 2014). We leveraged our access to patient derived xenografts from patients that have progressed on BRAF inhibitors to determine the molecular underpinnings of the MAPK-ER stress-autophagy signaling. We have identified a new step in this pathway that involves druggable targets, KSR2, SEC61, PERK, ATF4 (Ojha et al. Cancer Discovery 2019). We anticipate this work will uncover potential new combination therapies for BRAF mutant melanoma and other BRAF mutant cancers.
Announcement
- Aaron Kirchhoff joins the labs in 2025
- Anna Ramesh joins the lab as a Drexel co-op intern in 2025
- Drs. Vaibhav Jain, Amaravadi, Weissman and Mitchell receive the Melanoma Research Foundation Breakthrough Consortium Team Science Award in 2025 to work on novel mRNA-LNP therapy for melanoma RNA-LNP Therapeutics for Resistant Melanoma – Melanoma Research Foundation
- Suravi Ray, MS joins the lab in 2024
- Amanda Versace accepted to the Cell and Molecular Biology graduate program at the University of Pennsylvania
- Dr. Amaravadi was inducted into the Association of American Physicians in 2023
- Dr. Amaravadi becomes the Associate Director for Translational Research at the Abramson Cancer Center
- Lazlo Nziga joined the lab as a PASS student for the summer of 2021/2022 and was accepted into UPENN medical school
- Jennifer Lee was in the lab from 2018-2022 and was accepted to Johns Hopkins Medical School
- Dr. Amaravadi is the co-Director of the newly funded Wistar/UPENN SPORE in Skin Cancer. https://www.news-medical.net/news/20210922/Penn-Medicine-and-Wistar-secure-24117-million-SPORE-grant-for-three-melanoma-research-projects.aspx
- Cynthia Chude, former Lab member becomes starts an MDPhD program as the first Meharry-Wharton-Leonard Davis Institute scholar: https://ldi.upenn.edu/our-work/research-updates/new-wharton-school-meharry-college-joined-md-phd-program-welcomes-first-scholar/
Spotlight
 Anna Ramesh joined the Amaravadi Lab in March 2025 through Drexel University’s co-op program—a six-month, full-time professional experience that allows undergraduates to gain immersive, hands-on training in their field of interest. She is currently pursuing a Bachelor of Science in Biological Sciences at Drexel, where she is part of the BS/MD Early Assurance Program. Anna began her research journey in 2023 in the lab of Dr. Mauricio Reginato at the Drexel University College of Medicine, where she investigated lipid metabolism reprogramming in triple-negative breast cancer (TNBC). Her contributions toward developing brain-penetrant ACSS2 inhibitors that target breast cancer brain metastases (BCBM) led to a co-authored manuscript. She has presented her research on acetate’s protective role against ferroptosis in BCBM cells at national research conferences and earned multiple travel and enrichment grants for this work. In the Amaravadi Lab, Anna is exploring how inhibiting glycosphingolipid synthesis can enhance antitumor immune responses. She also shadows Dr. Ravi Amaravadi in clinic to gain exposure to oncology care in a translational research setting. Beyond the lab, Anna serves as the undergraduate representative on the Sidney Kimmel Board of Cancer Research Trainees, where she helps shape mentorship and training opportunities for early-career scientists across Philadelphia. Long-term, she hopes to bridge scientific discovery and clinical care to advance more precise and impactful approaches to cancer treatment.
Anna Ramesh joined the Amaravadi Lab in March 2025 through Drexel University’s co-op program—a six-month, full-time professional experience that allows undergraduates to gain immersive, hands-on training in their field of interest. She is currently pursuing a Bachelor of Science in Biological Sciences at Drexel, where she is part of the BS/MD Early Assurance Program. Anna began her research journey in 2023 in the lab of Dr. Mauricio Reginato at the Drexel University College of Medicine, where she investigated lipid metabolism reprogramming in triple-negative breast cancer (TNBC). Her contributions toward developing brain-penetrant ACSS2 inhibitors that target breast cancer brain metastases (BCBM) led to a co-authored manuscript. She has presented her research on acetate’s protective role against ferroptosis in BCBM cells at national research conferences and earned multiple travel and enrichment grants for this work. In the Amaravadi Lab, Anna is exploring how inhibiting glycosphingolipid synthesis can enhance antitumor immune responses. She also shadows Dr. Ravi Amaravadi in clinic to gain exposure to oncology care in a translational research setting. Beyond the lab, Anna serves as the undergraduate representative on the Sidney Kimmel Board of Cancer Research Trainees, where she helps shape mentorship and training opportunities for early-career scientists across Philadelphia. Long-term, she hopes to bridge scientific discovery and clinical care to advance more precise and impactful approaches to cancer treatment.