Mullins Lab
Research Overview
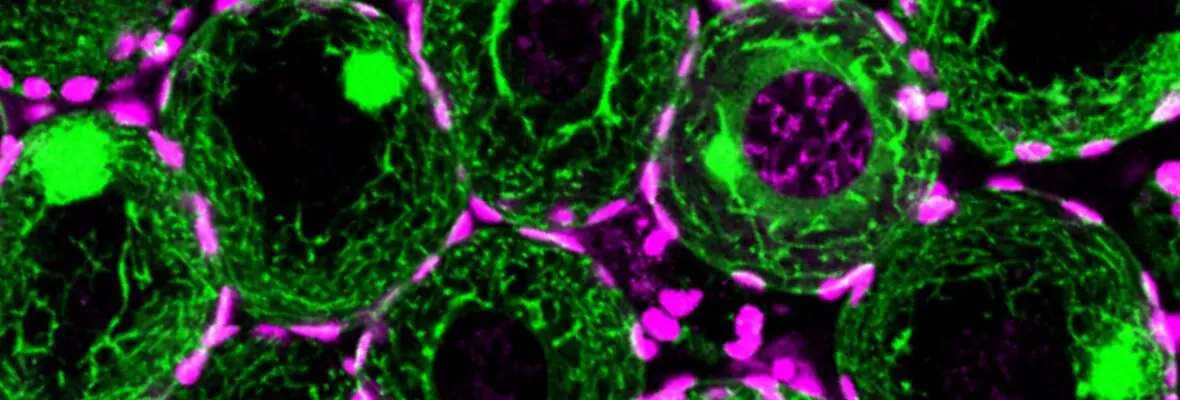
We are studying the molecular mechanisms by which a BMP (Bone Morphogenetic Protein) signal transduction pathway establishes different aspects of the vertebrate body plan. Various zebrafish mutants of BMP pathway components, as well as antisense knockdown approaches are used to dissect the molecular mechanisms by which this pathway establishes different cell types. We are studying the formation, function, and temporal regulation of a BMP activity gradient, which is implicated in specification of diverse cell types along the dorsal-ventral axis. We have shown that this gradient is essential in neural crest specification and is linked to dorsal-ventral patterning of neural tissue. Moreover, a subset of our defined components also function in post-embryonic heart development. Misregulation of BMP signaling leads to a debilitating disease in humans called fibrodysplasia ossificans progressiva (FOP). We are currently trying to establish a model for FOP in the zebrafish.
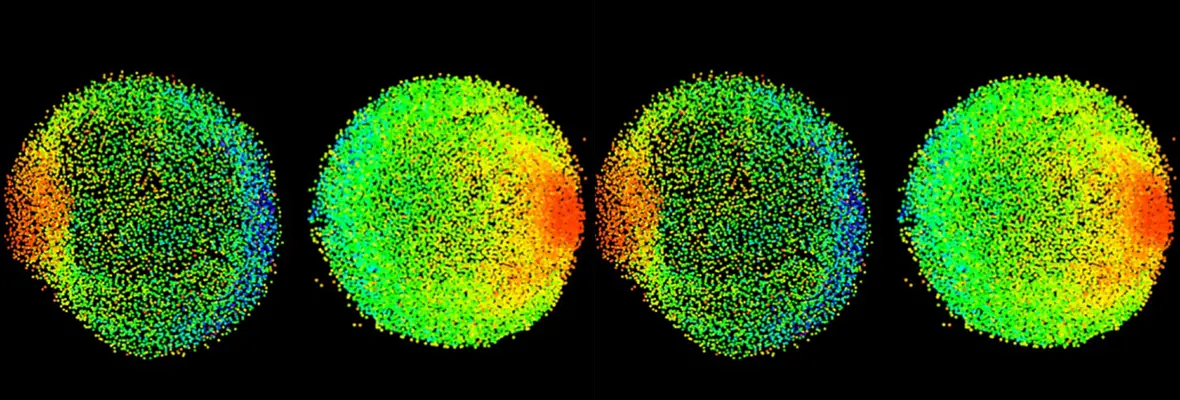
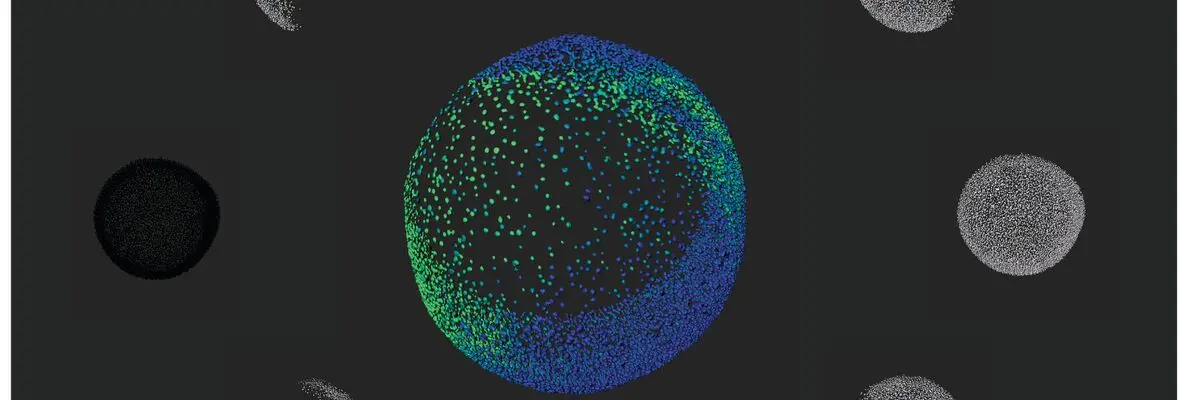
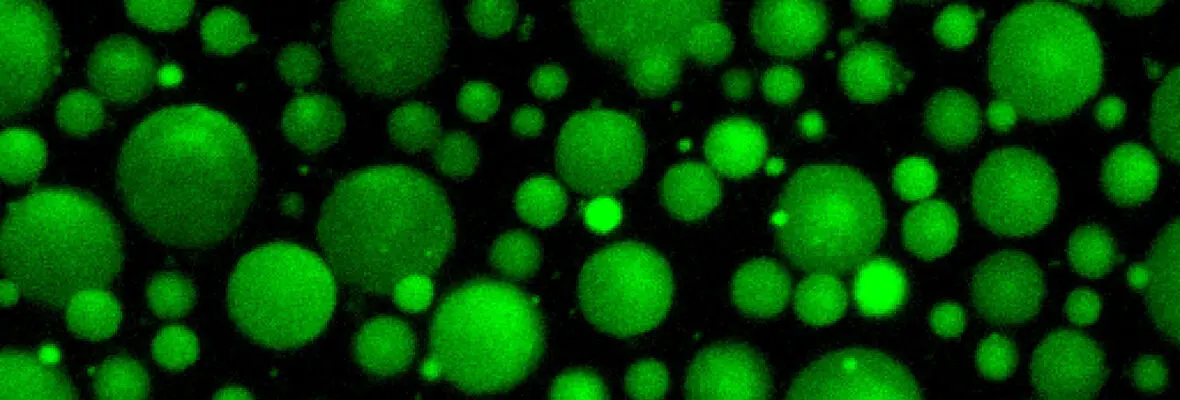
Elaboration of the vertebrate body plan relies not only on zygotic processes, but also on maternally-controlled processes: that is, processes that depend on products derived from the mother that are deposited into the oocyte and are critical for proper development of the embryo. To this end, we performed a large-scale maternal-effect mutant screen, not previously performed in a vertebrate, to identify mutants of key genes specifically required in the mother for oocyte development, egg activation, fertilization, the mid-blastula transition, and establishment of the axes of the vertebrate embryo. We identified numerous mutants in these processes and are studying the molecular and cellular basis for the defects, including positional cloning of the mutated genes.
Latest News
-
The Mullins Lab Went Fishing For Another Post Doc!
Monday, September 15, 2025
Big welcome to Dr. Satyam Srivastav, who is joining the Mullins Laboratory as a post doctoral fellow! He will be working on the piRNA pathway in zebrafish!
-
New Balbiani Body Buddy!
Monday, June 16, 2025
The Mullins Lab is very excited to welcome Avani Modak, a graduate student from the Developmental, Stem Cell, and Regenerative Biology graduate program! She will be investigating Balbiani body morphology and organelle dynamics during zebrafish oogenesis.
-
Allison Marvin and Megan Guerin Awarded F31 Fellowships from NICHD!
Tuesday, March 25, 2025
Congratulations to Allison and Megan! You can do any-fin you put your mind to! Allison will be investigating microtubule dynamics during egg activation, while Megan will be exploring the role of intrinsically disordered domains in Balbiani body and germplasm aggregation.