What does the study involve?
Our team is recruiting both women who have PMS/PMDD and women who don't have PMS/PMDD (healthy controls).
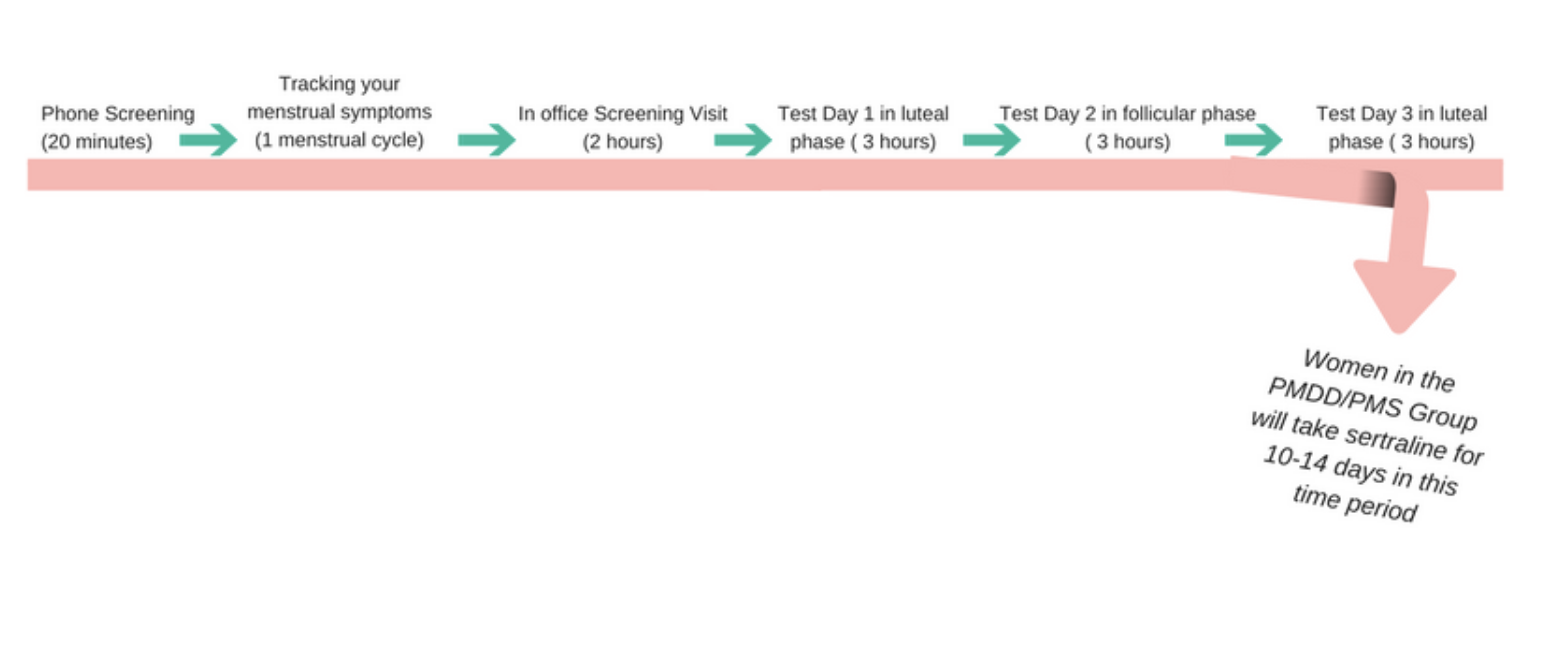
All women who participate in the study will complete 4 in-office visits over the course of 3-6 months. Each of these visits lasts 2-3 hours. This includes 1 screening visit where we clarify your eligibility, and 3 test days. At each of those 3 test days, we'll measure your response to stress in a few different ways.
Our team is also trying to see whether taking a low-dose of an FDA approved antidepressant medication prior to getting a period can decrease the severity of PMS or PMDD and change response to stress. All women who self-report symptoms of PMS or PMDD would take sertraline for the 10-14 days prior to getting their period in the final phase of the study. Healthy controls (women who do not experience PMS/PMDD) would not take medication.
To find out whether you would betin the PMS/PMDD group or the healthy control group of the study, we ask you to track your menstrual symptoms electronically. You would do this for 1 full menstrual cycle before enrolling in the study. Then, if you are eligible, you will continue to track these symptoms every day until you've completed the study.