Project 5: Chemoprevention of Asbestos-Inducing Lung Diseases
Significance
It has now been clearly established in both animal models and in humans that inhalation of asbestos fibers can lead to neoplastic diseases such as malignant mesothelioma (MM) and lung cancer. This is an important ongoing problem, since asbestos is still in used in many countries around the world and there is also a large pool of previously exposed individuals who are at risk for cancer. There is currently no effective screening or chemopreventive approach for these at-risk populations.
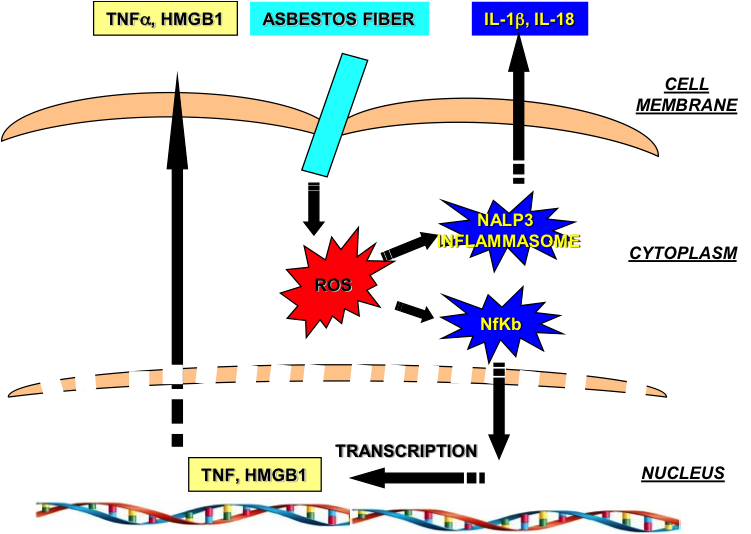
Given that recent studies have indicated that the pathogenesis of asbestos-induced cancers is due to chronic inflammation and oxidative tissue damage caused by persistent asbestos fibers, a well-tolerated and safe agent with anti-inflammatory AND anti-oxidant properties could thus potentially be used to prevent the development of MM in asbestos exposed populations. Based on preliminary findings, we have found that flaxseed lignans are safe, non-toxic compounds that have potent anti-inflammatory and anti-oxidant properties.
Hypothesis
We therefore hypothesize that that flaxseed or enriched flaxseed lignans could function as safe, non-toxic chemopreventive agents in asbestos-induced mesothelioma.
The goal of Project 5 is to provide data needed as the first steps to test and validate this hypothesis. To achieve our goal, we propose to:
- Evaluate the ability of the active lignan in flaxseed, Secoisolariciresinol diglucoside (SDG), to interfere with asbestos-induced ROS generation, inflammasome activation, and reactive oxygen species generation in macrophages and mesothelial cells in vitro.
- Evaluate the ability of wholegrain flaxseed or flaxseed-lignan complex given in diets to prevent acute asbestos-induced inflammation and oxidative damage in the mesothelial SV40 T-Antigen and the Nf2+/mut;Cdkn2a+/mut asbestos-induced malignant mesothelioma mouse models, and
- Evaluate the ability of wholegrain flaxseed or flaxseed-derived lignan SDG given in diets to prevent asbestos-induced MM formation and death in these same mouse models.
Project 5 will interact closely with three projects in this Superfund Proposal. Specifically, this will be accomplished by testing remediated asbestos from Project 1, working closely with the animal models of mesothelioma in Project 4 and taking advantage of the biomarker work in Project 6. If the studies show efficacy with safety, we will work closely with the Research Translation Core to implement application of our findings.
The natural extension of our work would be a toxicity and biomarker trial in which patients with heavy asbestos exposure who also had biomarkers of exposure (identified in Project 6) would be given flaxseed or the lignan formulation and changes in biomarkers identified. If we observed clear changes in this trial, wholegrain flaxseed (FS) or the purified flaxseed lignan component (FLC) enriched in the lignan SDG would be proposed to the NIEHS, NCI or EDRN for a larger chemoprevention trial in high-risk populations.
Project 5 will participate in the Interdisciplinary Research Training Core through the involvement of the postdoctoral fellows. This will provide the fellows with expertise in the application of environmental science and environmental health science aspects to issues related to Superfund hazardous waste and the potential adverse health effects that can result. Project 5 will also work closely with the Administrative Core for overall direction and for financial oversight. The Biostatistics Research Support Core will be used for chemoprevention study design and analysis of both efficacy and biomarker data. Finally, Project 5 will engage the Community Engagement Core to inform Stakeholder Advisory Group in Ambler on the potential efficacy of chemoprevention studies and work with the Research Translation Core to ensure that intellectual property related to the chemoprevention studies is secured.
Project Leaders
Melpo Christofidou-Solomidou, PhD, Project Leader
Research Associate Professor, Department of Medicine, Perelman School of Medicine, University of Pennsylvania
melpo@pennmedicine.upenn.edu
Steven M. Albelda, MD, Co-Project Leader
William Maul Measey Professor of Medicine, Department of Medicine, Perelman School of Medicine, University of Pennsylvania
albelda@pennmedicine.upenn.edu
Publications
Click Here to view all publications