Early Detection
The strongest argument for pursuing early detection of ovarian cancer lies in the stark contrast in survival rates between patients diagnosed at early versus late stages. Ovarian cancer, particularly high-grade serous carcinoma (HGSC), is notoriously difficult to detect early due to its lack of overt symptoms and the absence of a reliable screening test for the general population. However, evidence suggests that diagnosing ovarian cancer at an early stage significantly improves outcomes. The 5-year survival rate for patients diagnosed with ovarian cancer at an early stage (Stage I) is approximately 90%, compared to only 30-40% for those diagnosed at a late stage (Stage III or IV). Early-stage diagnoses typically mean that the cancer is confined to the ovaries or nearby tissues, where it is more treatable and manageable.
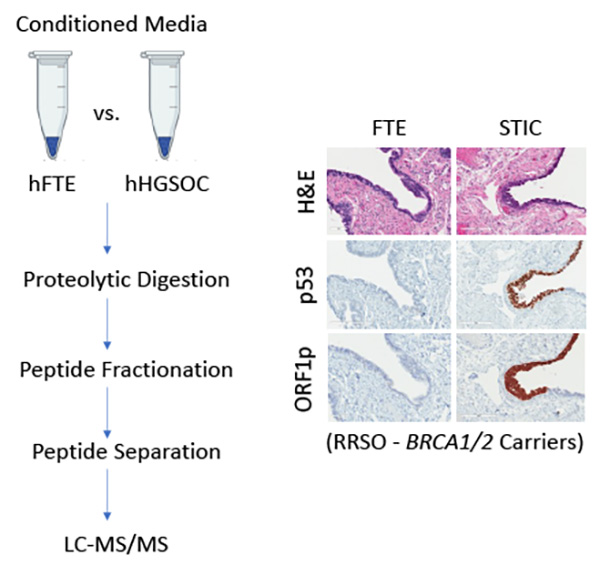
To enable early detection, we are applying mass spectrometry to interrogate the secretomes of malignant cells versus benign Fallopian tube counterparts in two tissue-proximal systems: (1) our Fallopian tube-derived ex-vivo cultures, and (2) tissue interstitial fluid (TIF) from matched fresh tissues. In addition to established biomarkers like CA125 and HE4, we have identified novel proteins, such as CRABP2 and LINE-1 ORF1p, that are secreted by cancer cells. LINE-1 ORF1p, in particular, represents a mobile genetic element typically silenced in normal cells but reactivated during neoplastic transformation. Ongoing research is focused on defining the translational utility and biological activity of these novel markers, which could provide valuable insights for early diagnosis and intervention.
Our lab is also making significant strides in leveraging the pre-cancer atlas we recently published, which combines high-plex imaging and spatial transcriptomics to map the molecular transitions from precancerous lesions to invasive HGSC. This atlas provides a rich dataset of early molecular and immune changes that can be leveraged to identify novel biomarkers for early detection. By integrating these findings with other 'omic' approaches, we aim to refine our understanding of the earliest stages of ovarian cancer progression and identify key molecular alterations that could serve as targets for early detection.
109dWith these cutting-edge technologies and approaches, we are advancing the development of non-invasive, reliable diagnostic tools that can detect ovarian cancer at its earliest, most treatable stages.
 1faf
1faf
