Research Interests
Cytoskeletal Motors
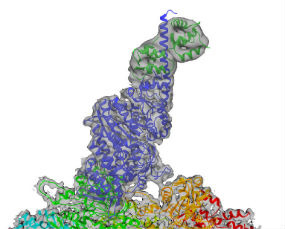 We are focused on investigating cytoskeletal motors (myosin, dynein, and kinesin). These remarkable nano-machines use chemical energy stored in our cells (in the form of ATP) to generate mechanical force and motion. Cytoskeletal motors are the engines that power muscle contraction, cell migration, intracellular transport, cell division, and cell shape. We are determining how these motors work at the molecular level, how they are physically connected to the machinery they are powering, how they are regulated, how they interact with other motors and signaling networks, and how their fundamental biophysical parameters impact cell function. We are using a range of biochemical, cell biological, single-molecule, and other biophysical techniques to better understand these proteins in health and disease.
We are focused on investigating cytoskeletal motors (myosin, dynein, and kinesin). These remarkable nano-machines use chemical energy stored in our cells (in the form of ATP) to generate mechanical force and motion. Cytoskeletal motors are the engines that power muscle contraction, cell migration, intracellular transport, cell division, and cell shape. We are determining how these motors work at the molecular level, how they are physically connected to the machinery they are powering, how they are regulated, how they interact with other motors and signaling networks, and how their fundamental biophysical parameters impact cell function. We are using a range of biochemical, cell biological, single-molecule, and other biophysical techniques to better understand these proteins in health and disease.
Motors in Muscle Contraction and Disease
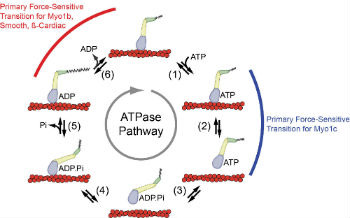 The beating of the heart is powered by the interaction of myosin motors with actin in muscle sarcomeres. Remarkably, myosin motors are able to inherently adjust their power outputs in response to mechanical loads. We are investigating mechanisms by which myosins power muscle contraction and change their contractile properties in response to mechanical loads. We are also investigating how mutations in myosins that cause human cardiomyopathies affect the ability of myosin to power cardiac contraction.
The beating of the heart is powered by the interaction of myosin motors with actin in muscle sarcomeres. Remarkably, myosin motors are able to inherently adjust their power outputs in response to mechanical loads. We are investigating mechanisms by which myosins power muscle contraction and change their contractile properties in response to mechanical loads. We are also investigating how mutations in myosins that cause human cardiomyopathies affect the ability of myosin to power cardiac contraction.
Cytoskeletal Assemblies, Membrane Transport, and Biomimetic Platforms

Molecular motors navigate and remodel complex cytoskeletal geometries that are important for their cellular function. We are investigating how motors bind and shape membrane substrates as they navigate the cellular geometries. Experiments include high-resolution tracking of intracellular motors and cargos. Additionally, we are using microfabrication techniques to build biomimetic cytoskeletal platforms to better understand the molecular interactions of the cytoskeletal with proteins and lipids that drive membrane transport and modeling.

