NEUROTRAUMA RESEARCH
The Smith Neurotrauma Laboratory focuses on the mechanical events at the time of brain injury and how they relate to long-term outcome. It specifically concentrates on the fate of nerve fibers in the brain, called "axons," that appear to be exquisitely vulnerable to trauma. Brain trauma is a devastating disease that affects over 2 million people in the United States each year. However, the mechanisms of brain trauma have only begun to be elucidated. Prior research has shown that some unique features of the physical damage induced by brain trauma can trigger progressive degenerative damage. This startling finding is the basis of our research efforts at Penn.
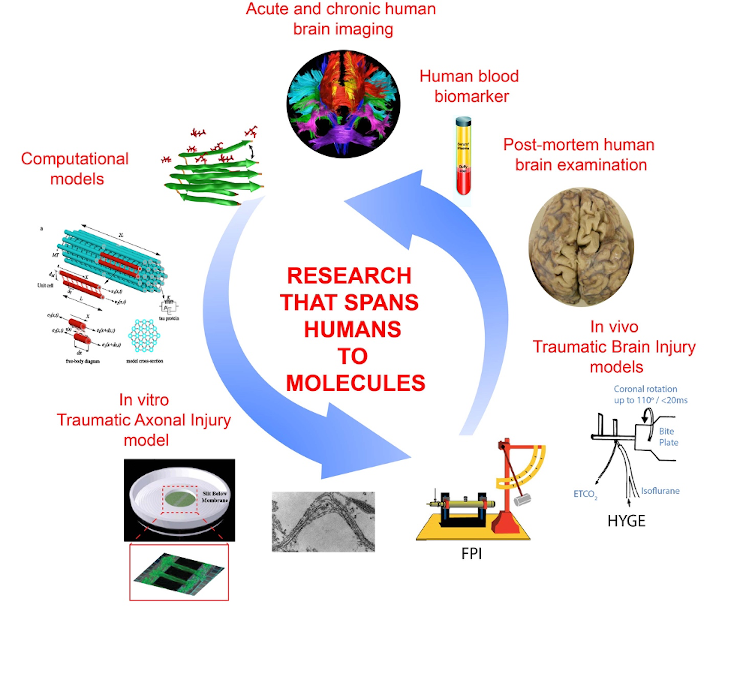
Paradoxically, the key features of the human brain that allow humans to design and drive automobiles are also their greatest liability in the event of a crash. With its massive size and high organization, the human brain can literally pull itself apart under the physical forces associated with traumatic brain injury. In particular, axons in the white matter appear poorly prepared to withstand damage from rapid mechanical deformation of the brain during trauma. Accordingly, axonal injury is one of the most common pathologies resulting from brain trauma, where the extent of axonal damage is thought to play a major role in functional outcome. Through a multidisciplinary approach which includes computer models, in vitro and in vivo experimental models, and clinical and post-mortem human data, research in the Smith Neurotrauma Lab aims to elucidate the mechanisms underlying axonal damage in traumatic brain injury.
NEUROREPAIR
While a primary strategy to repair spinal cord and other nerve injuries is to bridge the damage with axons, producing axons of sufficient length and number has posed a significant challenge. In the Smith Laboratory, scientists explore the ability of integrated central and peripheral nervous system (CNS and PNS) axons to grow long distances in response to continuous mechanical tension. Using a microstepper motor system, we have physically split an integrated neuron culture in two and progressively separated the two populations of neurons. The tension on the axons spanning the expanding gap induced "stretch-induced growth." Using this novel mechanism of axon growth, we are able to produce remarkably thick bundles of thousands of axons that can be grown to several centimeters in length. We propose that this technique may be exploited to produce transplant materials to bridge extensive nerve damage.




