Our Research
During cell division chromosomes must be distributed evenly between daughter cells. Errors in chromosome segregation constitute a serious threat to human health because they lead to abnormal genetic makeup, a hallmark feature of cancer cells. Our work aims to understand the mechanisms of accurate chromosome segregation during cell division. Specifically, we want to understand how spindle microtubules attach to kinetochore, a specialized chromosomal structure, and how these attachments drive chromosome motions that ultimately achieve accurate inheritance of genetic information. In our experimental work we use highly advanced biophysical approaches, such as single-molecule fluorescence microscopy and laser tweezers, to study the interactions between isolated kinetochore proteins and dynamic microtubules in vitro under conditions that recapitulate aspects of normal chromosome-microtubule attachment in cells. Current research is focused on the kinesin motor CENP-E, microtubule-binding protein complexes Ndc80, Dam1 and Ska1, and the regulatory kinase Aurora B. With mathematical models we seek to develop a quantitative, mechanistic description of how these proteins interact with microtubules and function at the kinetochore interface, linking specific molecular characteristics with physiological behavior of mitotic cells.
1. Kinetochore motors: kinesin CENP-E
CENP-E is the major plus-end directed kinetochore motor, which transports chromosomes from spindle pole to spindle equator. We have recently discovered that in addition to this transporting role, CENP-E kinesin can also associate processively and bi-directionally with the tips of dynamic microtubules in vitro. Our working model is that in mitotic cells the CENP-E motor contributes to the end-on kinetochore-microtubule attachments. The ongoing work is aimed to recapitulate these interactions using purified components in vitro and to gain deep mechanistic understanding of CENP-E activity and regulation.
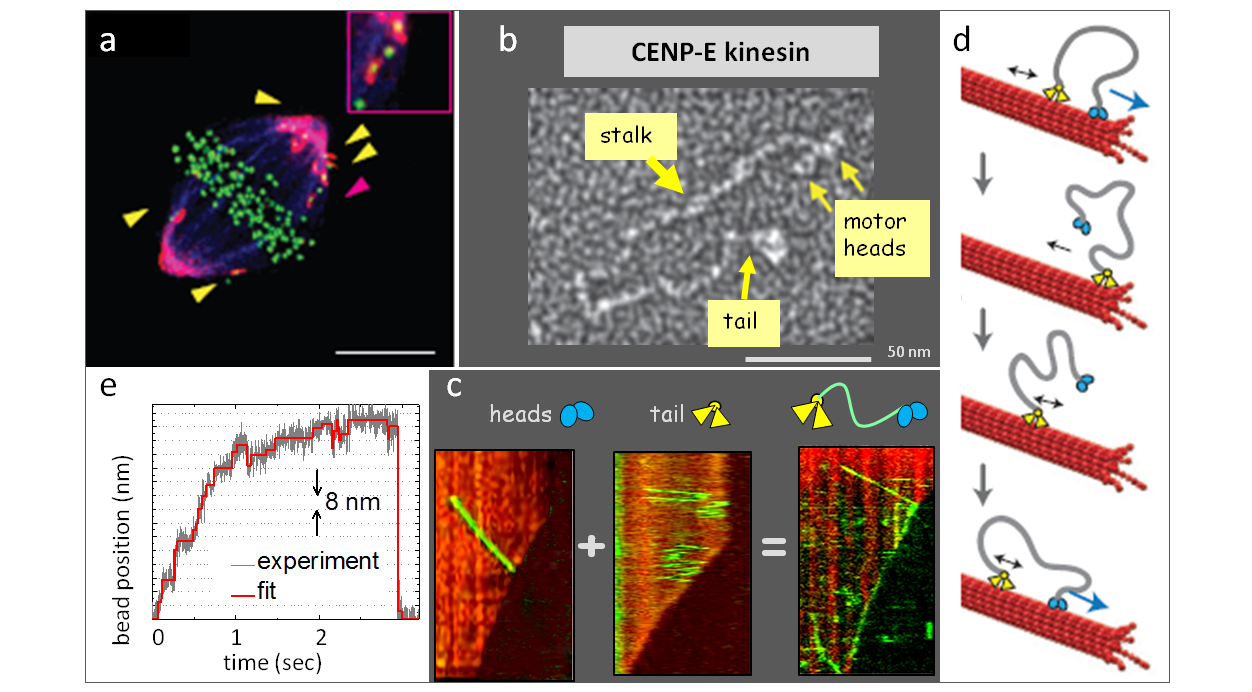 Legends to images: a. Immunofluorescent image of a mitotic cell with inhibited CENP-E motor. b. Electron micrographs of the highly elongated kinesin CENP-E (image from Kim et al., 2008). c. Kymographs of the motions of CENP-E protein and its motor and tail domains on dynamic microtubules. d. Model for bi-directional processive CENP-E tip-tracking. e. Stepping of a bead-attached CENP-E in an optical trap.
Legends to images: a. Immunofluorescent image of a mitotic cell with inhibited CENP-E motor. b. Electron micrographs of the highly elongated kinesin CENP-E (image from Kim et al., 2008). c. Kymographs of the motions of CENP-E protein and its motor and tail domains on dynamic microtubules. d. Model for bi-directional processive CENP-E tip-tracking. e. Stepping of a bead-attached CENP-E in an optical trap.
|
Video 1 |
Video 2 |
Video 3 |
Legends to videos: Video 1. Motor heads of CENP-E fall of the dynamic microtubule tip. Video 2. Full length CENP-E molecule tracks bi-directionally with dynamic microtubule tip. Video 3. Mathematical model of bi-directional tip-tracking by the “tethered motor” mechanism.
References:
Gudimchuk et al, (2013) Kinetochore kinesin CENP-E is a processive bi-directional tracker of dynamic microtubule tips. Nature Cell Biol., 15(9): 1079-88.
Vitre et al., (2014) Kinetochore-microtubule attachment throughout mitosis potentiated by the elongated stalk of the kinetochore kinesin CENP-E. Mol. Biol. Cell 25(15):2272-81.
Barisic at al, (2015) Microtubule detyrosination guides chromosomes during mitosis. Science 348(6236): 799-803.
2. Coupling to the dynamic microtubule end via the kinetochore MAPs
Conventional ATP-dependent motors are important for the fidelity of chromosome segregation but they are dispensable for pole-directed chromosome motion in dividing cells, suggesting that disassembling microtubules can serve as a minus-end-directed motor. In vitro, depolymerizing microtubules can move objects that are appropriately coupled to their shortening ends. We hypothesize that a similar mechanism plays central role in the pole-directed chromosome movement and that specialized, kinetochore-localized macromolecular couplers capture the energy from tubulin disassembly, while simultaneously providing stable attachments to the shortening microtubule ends. For example, microtubule associated protein (MAP) complex called Dam1 can form a ring around the microtubule, which is thought to provide strong coupling to dynamic microtubule ends in yeast cells. However, the force-bearing coupling mechanism at human kinetochore is still ill-defined. Our current work examines human kinetochore Ska1 complex, which may function analogously to yeast Dam1 protein.
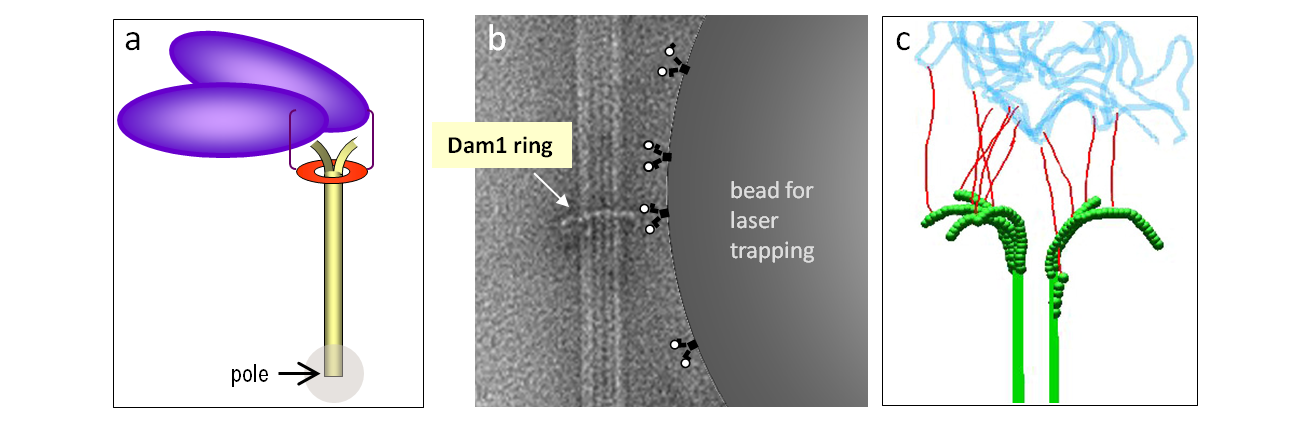 Legends to images: a. Schematic for the ring-dependent kinetochore coupling. b. Electron micrograph of Dam1 ring and a drawing of a laterally attached microbead, which is used in laser trapping experiments. c. Reconstruction of kinetochore microtubule ends in mammalian cells suggests the importance of elongated fibrillar attachments.
Legends to images: a. Schematic for the ring-dependent kinetochore coupling. b. Electron micrograph of Dam1 ring and a drawing of a laterally attached microbead, which is used in laser trapping experiments. c. Reconstruction of kinetochore microtubule ends in mammalian cells suggests the importance of elongated fibrillar attachments.
|
Video 1 |
Video 2 |
Video 3 |
Legends to videos: Video 1. Alexa488-labeled Dam1 protein complex tracks with microtubule depolymerization, which was induced by a photoablation of the stabilizing, rhodamine-labeled microtubule cap. Video 2. Mathematical model for Dam1 ring tip-tracking by the forced walk mechanism. Video 3. GFP-labeled Ska1 complex tracks with the depolymerizing microtubule end.
References:
Volkov et al., (2014) Preparation of segmented microtubules to study motions driven by the disassembling microtubule ends. J. of Vis. Exp., 15(85).
Volkov et al., (2013) Long tethers provide high-force coupling of the Dam1 ring to shortening microtubules. Proc. Natl. Acad. Sci. (USA) 110(19): 7708-13.
Schmidt et al., (2012) The kinetochore-bound Ska1 complex tracks depolymerizing microtubules and binds to curved protofilaments. Dev. Cell 23(5): 968-80.
Grishchuk et al., (2011). Force generation by dynamic microtubule polymers. In Comprehensive Biophysics, Vol 4, Molecular Motors and Motility. Oxford: Academic Press, pp. 93-117.
Welburn et al., (2009) The human kinetochore Ska1 complex facilitates microtubule depolymerization-coupled motility. Dev. Cell 16(3): 374-385.
Grishchuk et al., (2008) The Dam1 ring binds microtubules strongly enough to be a processive as well as energy-efficient coupler for chromosome motion. Proc. Natl. Acad. Sci. (USA) 105(40): 15423-8.
Grishchuk et al., (2008) Different assemblies of the DAM1 complex follow shortening microtubules by distinct mechanisms. Proc. Natl. Acad. Sci. (USA) 105(19): 6918-23.
McIntosh et al., (2008) Fibrils connect microtubule tips with kinetochores: a mechanism to couple tubulin dynamics to chromosome motion. Cell 135: 322-33.
Efremov et al., (2007) In search of an optimal ring to couple microtubule depolymerization to processive chromosome motions. Proc. Natl. Acad. Sci. (USA) 104(48): 19017-22.
Grishchuk et al., (2005) Force production by disassembling microtubules. Nature 438: 384-388.
3. Quantitative approaches to study kinetochore-microtubule interface
The connection between kinetochore proteins and microtubules are highly stochastic, and they can be tuned by regulatory enzymes, like Aurora B kinase. Using quantitative fluorescent microscopy approaches in vitro and with the help of molecular dynamics simulations, we study the biophysical mechanisms that underlie interactions between kinetochore proteins and microtubules and how these interactions are regulated by phosphorylation. These experimental data guide our multi-scale models of kinetochore-microtubule interface, revealing which aspects of mitotic physiology can be attributed to the specific kinetochore proteins.
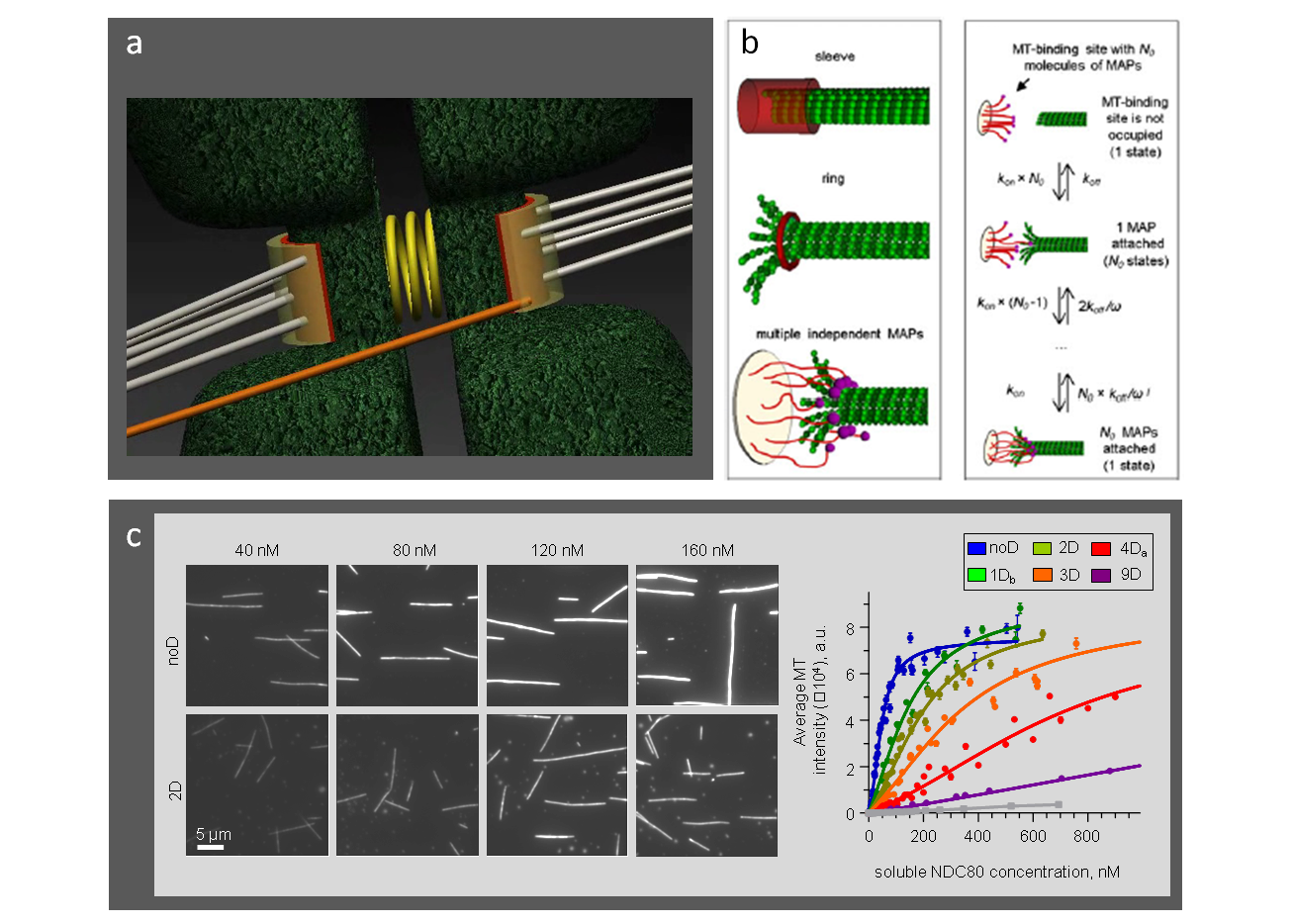 Legends to images: a. Schematic of the microtubule-binding interfaces at sister kinetochores. b. Different biophysical designs for microtubule interactions with kinetochore proteins. c. Experimental microtubule-binding curves for different Ndc80 phosphomutants provide the kinetic constants for mathematical modeling
Legends to images: a. Schematic of the microtubule-binding interfaces at sister kinetochores. b. Different biophysical designs for microtubule interactions with kinetochore proteins. c. Experimental microtubule-binding curves for different Ndc80 phosphomutants provide the kinetic constants for mathematical modeling
|
Video 1 |
Video 2 |
Video 3 |
|
Video 4 |
Video 5 |
Video 6 |
Legend to video: Video 1. Model representation of the microtubule binding interface. Video 2. Stochastic model for microtubule-binding to the molecular lawn with the phosphorylation-controlled Ndc80 complexes. Video 3. Single unphosphorylated NDC80-GFP complexes bind and diffuse along the taxol-stabilized microtubules. Video 4. Molecular dynamics simulation of the NDC80-microtubule interface with unphosphorylated Hec1 tail. Video 5. Molecular dynamics simulation of the NDC80-microtubule interface analogous to Video 4 but with Hec1 tail containing 9 phosphomimetic substitutions (purple spheres). Video 6. Simulation of the sister kinetochore interactions with dynamic microtubules in a cell recovering from the monastrol-like block.
References:
Zaytsev, A.V., Grishchuk, E.L. (2015) Basic mechanism for biorientation of mitotic chromosomes is provided by the kinetochore geometry and indiscriminate turnover of kinetochore microtubules. Mol. Biol. Cell., 26(22):3985-98.
Zaytsev, A.V., Mick, J.E., Maslennikov, E., Nikashin, B., DeLuca, J.G., Grishchuk, E.L. (2015) Multisite phosphorylation of the NDC80 complex gradually tunes its microtubule-binding affinity. Mol. Biol. Cell., 26(10):1829-44.
Zaytsev et al., (2014) Accurate phosphoregulation of kinetochore-microtubule affinity requires unconstrained molecular interactions. J. Cell Biol., 206(1):45-59.
Zaytsev et al., (2013) Highly transient molecular interactions underlie the stability of kinetochore-microtubule attachment during cell division. Cell. Mol. Bioeng., 6(4).
4. Atomic scale and molecular-mechanical microtubule modeling
Microtubules are essential for cell division and they play vital roles in maintaining cell polarity and architecture, as well as during intracellular transport. We explore the biomechanical and dynamic properties of these polymers using atomic-scale modeling, molecular dynamics simulations and molecular-mechanical models. With these approaches we hope to gain deep mechanistic understanding of the force generation by depolymerizing microtubules and the nature of microtubule catastrophes, as well as to assist development of novel microtubule-disrupting and anti-tumor drugs.
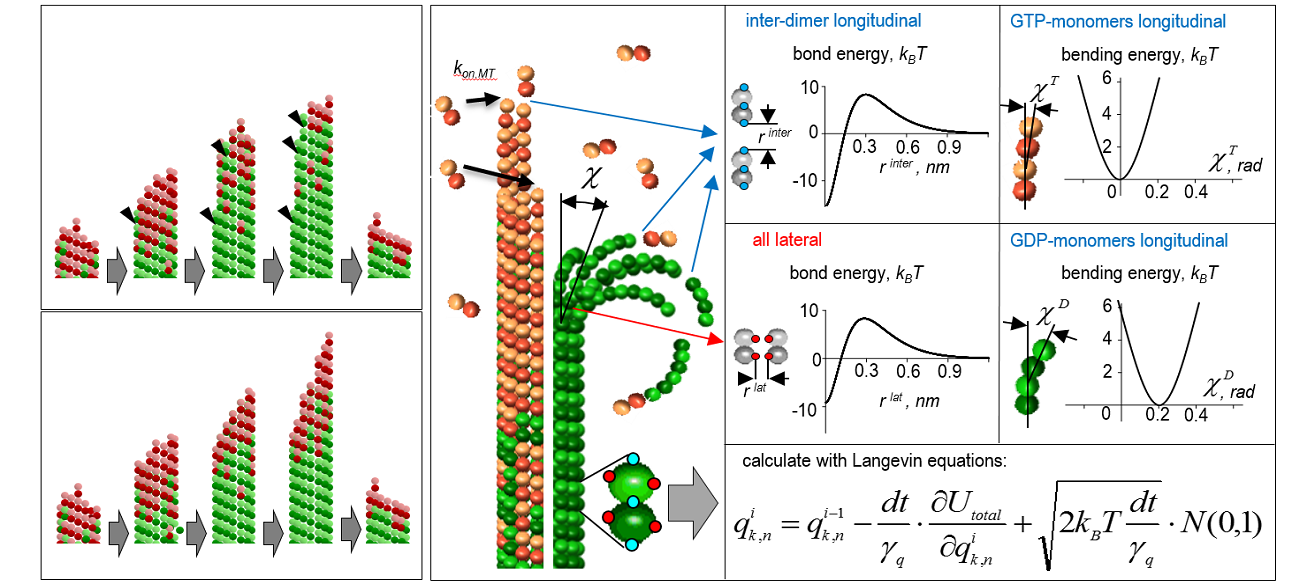 Legend to image: Molecular-mechanical model of the dynamic microtubule.
Legend to image: Molecular-mechanical model of the dynamic microtubule.
|
Video 1 |
Video 2 |
Video 3 |
Legend to video: Video 1. Microtubule nano-indentation in silico. Video 2. Dynamic microtubule growth, catastrophe and shortening calculated using ~ 20-fold accelerated GTP hydrolysis rate. Video 3. Simulated “dilution” experiment, in which microtubule first elongates in the presence of soluble tubulin. Soluble tubulin is then removed, causing the microtubule to pause and eventually depolymerize.
References:
Zakharov, P., Gudimchuk, N., Voevodin, V., Tikhonravov, A., Ataullakhanov, F. I., Grishchuk, E. L. (2015) Molecular and mechanical causes of microtubule catastrophe and aging. Biophysical J. 109(12):2574-91.
Kononova et al., (2014) Tubulin bond energies and microtubule biomechanics determined from nanoindentation in silico. JACS 136: 17036−45.
Molodtsov et al., (2005) A Molecular-mechanical model of microtubules. Biophys. J. 88: 3167-79.
Molodtsov et al., (2005) Force production by depolymerizing microtubules: a theoretical study. Proc. Natl. Acad. Sci. (USA) 102: 4353-8.