Recruitment: Strategies, Resources, Privacy
Privacy Considerations, Covered Entity while Recruiting
Research is one of Penn Medicine’s core missions. Patients are informed of this mission, the ability to use and disclose PHI under oversight by IRBs, and the choices to opt out of Research contact and health information exchange, through the Notice of Privacy Practices or NPP
Patients are able to indicate their choice to be opted out of being contacted for research recruitment in their medical record.
They can do this by asking their front desk staff to indicate this on their chart, log into My Penn Medicine –Penn Medicine’s patient portal, call the Privacy Office or the Office of Clinical Research.
We recommend that opt out language be included in all recruitment communications.
Covered Entity - Impact on Recruitment
A covered entity under HIPAA is a health plan, a health care clearinghouse, or a health care provider that electronically transmits protected health information.
At Penn Medicine, the covered entity includes the hospitals that are under the University of Pennsylvania Health system or UPHS AND the Perelman School of Medicine or PSOM.
Hospitals include HUP, Penn Presbyterian, PAH, Chester County, Princeton Health, Lancaster General. Other schools that are part of the University of Pennsylvania such as Nursing School, Dental School, Wharton, Engineering, Arts and Sciences, Annenberg etc. are NOT part of the covered entity.
Private Health Information OR PHI, and access to systems that house PHI, for example, the medical record system; is provided only to Penn Medicine employees. All other university employees or external collaborators that would like to use data systems, must request through a sponsor within the covered entity.
Recruitment Strategies & Resources
These have been categorized into the following methods. Best practices and tips have been listed for each method.
Online
This method allows patients to search for, reach out to research teams, and inquire about participating in clinical research at Penn Medicine.
1. List research study/ trial on Penn Medicine’s Clinical Research Finder site, Penn iConnect
-
Benefits are: HIPAA compliant, free of cost, study can have its own page, platform is public facing, allows patients and public, including non-Penn physicians to search for research studies, and send email inquiries to teams.
-
Add pre-screeners, tracked links for social media ads, use research volunteer registry with 10,000+ volunteers. Go to Penn iConnect
2. Research teams can list their studies on the Department, Center, or institutes website
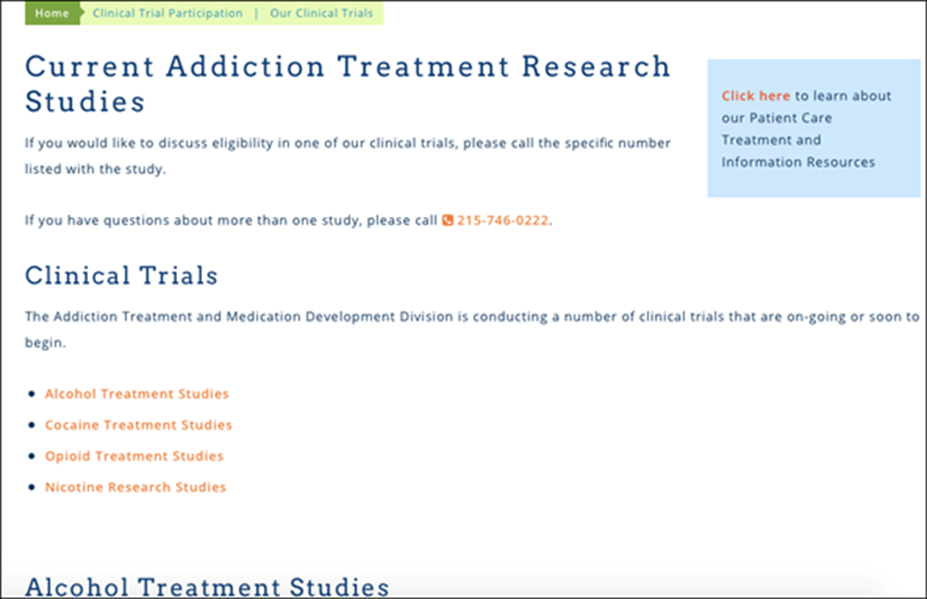
3. Similarly, you could list the study on the PI/ physicians profile on Penn Medicine’s website. You would contact your department’s websites Web Master or owner, or Penn Medicine IS to get the study listed.
Print advertisements/ Mixed Media
This includes flyers, local ads in newspapers, clings on public transportation, broader outreach using web podcasts
1. Research teams can use the Penn iConnect Patient recruitment management system to create tracked with QR codes, on their flyers, at no cost.
2. Local Philadelphia newspapers such as Tribune and Al Dia like to advertise about clinical research thru their print and digital newspapers. More details on cost are listed in the Penn Manual for Clinical Research
- Advertising through SEPTA and Indego bike share- Large outreach can be done using clings on SEPTA, Indego the bike share company.
-
These large visual outreach options are typically expensive. It’s advisable to use these options to create large scale outreach for a research program, department or initiative, versus a single research study or trial.

3. CureTalks@Penn is a Research interview podcast service offered through the Office of Clinical Research.
-
These interviews help research teams conduct wider outreach about their research trials or initiatives, or spread the word for hard to recruit, rare disease trials.
-
These interviews are done at no cost to the Penn research team
-
The interview is done typically with the study PI, 2 -3 subject experts. The interview is conducted virtually, edited and placed on Penn Medicines website and social media platforms, and YouTube
-
If interview done to aid recruitment for specific trial, interview script requires IRB approval.
In-clinic recruitment
Is where patients are approached at point of care. These typically happen in a clinic setting, in a hospital, doctors office, emergency room, waiting rooms
1. Physician Referrals (within and related departments)
-
Physician investigators are able to refer their own patients to other physicians in practices at Penn medicine if they know of open studies.
-
You could walk over with a copy of the IRB approved flyer, summary sheet or consent form to hand to the patient if they would like to look through and decide later.
-
Research teams can use Dear Physician, Dear Patient emails or letters templates. Once the research team and the PI establish contact you may receive approval from the physician providing care, to reach out to the patient
-
Review PHI and email guidance before communicating with patients via email. These guidance documents are titled: Avoid and Minimize PHI in Email and Use of Email During the Conduct of Research
2. Network with other practices and clinics
Research teams networking with departments and physicians that directly provide care for your protocol’s target population. Teams may also work with clinical department practice managers to get permission, and place flyer stands, place screensavers in TVs at patient waiting areas.
3. Electronic Medical Record (EMR) -PennChart
-
EPIC/PennChart allows notifying physicians that their patient may be potentially eligible for a research study is by using Best Practice Advisories (BPAs)
-
These would be limited to a team of physicians that are familiar with the study, and have been notified of potentially receiving notifications
-
Teams may also use research report templates in Pennchart, such as the “Upcoming Appointment search” report to get a sense of whether you may have potential participants coming in for clinic visits.
-
Various available patient recruitment options using PennChart/ EPIC are listed in this slide deck.
-
Contact the PennChart Research team for demos or consults of these functions.
-
Then submit your request to IS Service Desk to get started on technical build for your protocol.
4. Patient Recruitment Management System – Penn iConnect
In-clinic patient addition feature allows team to enter, track and pre-screen patients from clinic / point-of-care. More on our Patient Recruitment Management System (PRMS) page.
Email, Mail, Patient Portal, Phone
-
Dear Physician, Dear Patient emails, letters- these letters or emails can be used as an introduction to a patient at Penn Medicine, by someone already known to them.
-
Teams should consider adding other recruitment materials such as brochures, flyers, QR codes, along with the latter to provide more information about the study.
-
Remember to submit this letter/ email to the IRB, for approval, before use.
-
Emails to potentially eligible patients can be sent using the Electronic Medical Record, PennChart, through the patient portal, called myPennMedicine recruitment. Navigate to Recruitment via myPennMedicine
-
Some considerations while emailing or sending a letter to patient to maintain privacy are:
-
Add these to your letter/ email: introduction (PIs name, Penn Medicine, Department), goal of study, link/QR code/ brochure for more information, basic eligibility, brief description of assessments (survey, blood, draw, MRI), location of study or virtual, compensation if any.
-
Provide Opt-out option in the communication (both for your study and overall recruitment contact).
-
DO NOT include any PHI in any email communications. Email is NOT secure.
-
-
Phone (cold-calls- allowed but least encouraged).
Social Media
-
All clinical trial and research study social media ads must be placed on Penn Medicine-Clinical Research pages (Facebook, Instagram)
-
Free social media posts can yield outreach, for wider reach don’t yield effective recruitment- even $25 goes a long way.
-
When creating a campaign, create ads and posts around a larger idea, not single trial- Women’s Health, HIV awareness, Rare Diseases
-
Social Media ads must be created in conjunction with other online recruitment strategies.
-
Review social media guidance by Penn IRB and Marketing HERE
-
To put in free post or a paid ad on these social media pages, teams must complete training and get Business Manager access. See how to HERE
-
Central pages managed by Office of Clinical Research and/ or Penn Medicine Marketing are:
Mobile Apps
Mobile technologies allow research teams to reach participants and conduct parts or all of their study remotely.
Benefits include:
-
Remote consent or eConsent
-
Remote data collection or ePRO (Patient-Reported Outcomes)
-
Quick communication
-
Quicker appointment scheduling, change
-
Enhanced symptom monitoring and tracking
-
Quicker decision-making ability
Challenges / considerations include:
-
Lack of planning and team set up
-
Budget constraints
-
Complexity of integrations
-
Maintaining engagement with participants
-
Reliance on access to technology as a barrier to recruitment
Here are a list of mobile app technologies and vendors that are vetted by Penn Medicine IS, and allow quicker start times.
-
XPAND: Link to vendor website HERE. Link to related resources on this website HERE
-
Way2Health: Link to product’s website HERE
-
Telehealth/ Telemedicine: Use of remote visits for research subjects have allowed research to continue during through the COVID19 pandemic, and have proven to be an effective and satisfying tool for the participant in research.
-
Use of these platforms are more for post-consent visits, they could be used to discuss the consent of the protocol, but may not be used for cold- call recruitment.
-
The general Penn Medicine Telemedicine guidance provides a broad overview of different platforms that may be used for virtual research visits.
-
See more information in Policies, Procedures, Guidance section of this website; under Telemedicine
If you are in need of recruitment and retention consultation, please contact the Office of Clinical Research , CC: Maha Dutt or Tom Hohing (mdutt@upenn.edu, thohing@upenn.edu). Choose between Mondays (10:30-12:30), or Fridays (12-1:30pm).