Welcome to the Beers Laboratory
The Beers' Laboratory for Lung Epithelial Biology in the Pulmonary and Critical Care Division in the Department of Medicine is housed in state of the art wet bench space in the Penn-CHOP Lung Biology Institute in Edward J. Stemmler Hall located in the heart of the Perelman School of Medicine campus at 3450 Hamilton Walk, Philadelphia, PA 19104. Originally constructed in 1978 and completely renovated for the science of the 21st century, Stemmler Hall has over 250,000 sq feet of facilities for world class research, teaching, and animal care. Its strategic juxtaposition between the Hospital of the University of Pennsylvania, Children’s Hospital of Philadelphia and many of PSOM’s major research buildings facilitates collaborative investigations in basic and translational biomedical research.
Our lab is composed of a highly collaborative team that includes a physician scientist, fundamental PhD staff scientists, postdoctoral fellows, and undergraduate students. In addition to trainees, the lab is supported by a group of highly skilled research technicians to enhance the research enterprise and provide administrative support accelerating research discovery and innovation. We encourage you to explore our lab members page as many former students and postdocs from our lab are emerging as the next generation of leaders in pulmonary research and medicine.
Research Portfolio of the Lung Epithelial Biology Laboratory
Our group is dedicated to the characterization of cellular and molecular mechanisms underlying the biology of the distal lung epithelium in health and its role in the pathogenesis of lung disease with a particular focus on Idiopathic Pulmonary Fibrosis (IPF) and Children’s Interstitial Lung Disease (chILD).
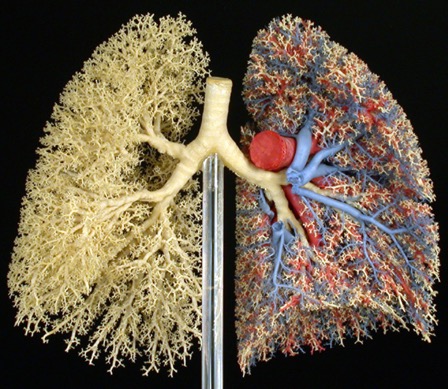
The alveolar type II pneumocyte (hereafter alveolar type 2 cell (AT2)) is an endodermally derived, multifunctional, polarized epithelial cell spatially restricted to the distal mammalian adult lung (Figure 1). Since the first report of the successful isolation of a surfactant producing epithelial cell from the lungs of rodents (Kikawa and Yoneda 1974 ), the major function of AT2 cells have been defined as the primary cells for synthesis and secretion of a surface-active film of biochemically heterogeneous lipoprotein mixture (lung surfactant) that reduces surface tension at air-liquid interfaces and allows for maintenance of alveolar stability at low lung volumes (Figure 2). Beyond playing a major role in surfactant homeostasis in the distal lung, there have been a number of other functional, molecular, and metabolic activities associated with an AT2 phenotype. These include but are not limited to a role in alveolar ion transport and fluid balance, innate host defense, and very importantly as a progenitor population for lung repair being capable of both proliferative expansion (self-renewal) and of transdifferentiating to type 1 (AT1) epithelial cells.
Recent Lab News!
We have some wonderful news updates!
1. Congratulations to Dr. Aditi Murthy and Dr. Sarah Bui for their recent publications!

2. Congratulations to Dr. Luis Rodriguez for embarking on his new journey of starting his own lab at George Washington University! Information about The Rodriguez Lab is linked here, please check it out! More information can be found in his bio on the "Where Are They Now?" page.
Publication Spotlight
- Divergent pathways of surfactant protein C maturation for disease-associated isoforms Sunday, March 1, 2026
- Regulatory T cells protect against aberrant remodeling in a mouse model of pulmonary fibrosis Saturday, December 27, 2025

