Achilles Tendinopathy Research Project 1
Current treatment strategies for Achilles tendinopathy seek to modulate the tendon mechanical environment to promote healing and regeneration. However, a lack of fundamental understanding of tendinopathic mechanobiology limits optimization of rehabilitation strategies and impedes development of targeted mechanotherapeutics. This project will address these gaps using orthogonal experiments across human tendinopathy, rat overuse, and mechanistic mouse models. The proposed studies will establish a framework to inform mechanotherapeutic strategies and identify novel signaling targets. Tendinopathy, particularly caused by overuse,
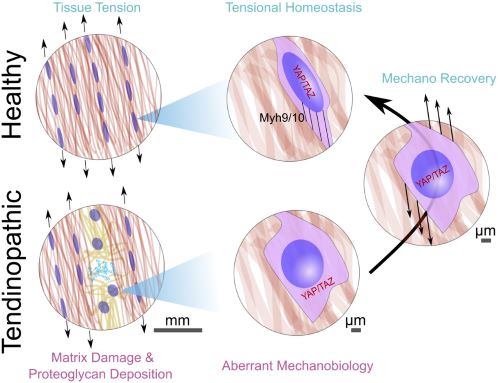
often results in tissue microdamage. This microdamage, in turn, impairs transmission of tensile mechanical signals through the tissue, resulting in under-loading of endogenous tendon cells. Our preliminary data show that tendon cells exhibit a biphasic response to tendon de-tensioning. De-tensioning causes a transient loss in cytoskeletal tension and mechanotransductive signaling, resulting in upregulation of matrix degrading enzymes like matrix metalloproteinases. Subsequently the tendon cells attempt to re-tension the matrix by cytoskeletal contraction and matrix re-organization. This biphasic response represents a novel framework for understanding the development, progression, and translational intervention of tendinopathy. In this study, we will 1) determine how tendinopathic disease impacts Achilles tensional homeostasis across the spectrum of disease, 2) define the mechanotransductive mechanisms, and 3) determine how rehabilitative loading restores Achilles tensional homeostasis for translational impact. Using our expertise in tenogenesis and tendinopathy (Dyment) and mechanobiology (Dyment/Boerckel), this translational project will provide a comprehensive understanding of the mechanotransductive mechanisms that drive aberrant and chronic matrix remodeling during Achilles tendinopathy, define mechano-response benchmarks in controlled assays that may predict clinical patient outcome, and identify potent drug targets for the next generation of tailored mechanotherapeutics to complement the current gold standards of physical therapy and surgery.








