Alzheimer's Disease Therapeutics Accelerator Projects
The IOA's first round of funded Alzheimer's Disease Therapeutics Accelerator Projects have been announced!
The University of Pennsylvania Institute on Aging (IOA) is pleased to announce a Letter of Intent (LOI) request for collaborative projects that focus on developing next-generation therapies for Alzheimer’s disease (AD). These projects will be supported by a research grant obtained by the co-Directors of the IOA from the Delaware Community Foundation.
Alzheimer's Disease Therapeutics Accelerator Projects
Eligibility
This award will support projects that focus on AD therapeutics including projects that address comorbid pathologies in the setting of Alzheimer’s disease. Both basic science and translational projects are eligible. However, basic science projects will only be considered if they focus on therapeutic development; projects that focus on mechanisms of disease without work towards a therapeutic will not be supported. Biomarker projects will be considered only if they describe evidence that they are directly related to enhancing existing or novel therapeutics.
Leaders for these proposed projects may reside in any of Penn’s 12 schools and priority will be given to projects that work across labs. Applicants are strongly encouraged to include a patient-facing clinical researcher to enhance the likelihood of future translation. Applicants who are IOA members will receive special preference and all applicants are encouraged to become members. Select teams of investigators will work collaboratively with the IOA to further develop their project(s).
Funding
Each project may request funds for up to $250,000 in total costs (direct costs will be $227,300 after a 10% indirect cost is applied). We anticipate that funds will be used over 1 to 2 years. Exceptional projects may request additional funding after the initial funds have been spent. Salary support for Project Leaders should be commensurate with their effort needed to execute the project.
Application Process
Letters of Intent (LOI) can be up to 1 page long and must include:
- Name(s) of the Project Leader(s) with contact information (including email address)
- Title of proposed project
- Overarching goal of project
- Abstract OR one paragraph description of the project
- Explanation of the project’s potential to result in or enhance AD therapeutics
LOIs will be accepted on a rolling basis and must be emailed to Kathy Jedrziewski, IOA Deputy Director at jedrzmk@pennmedicine.upenn.edu. We anticipate funding several projects through 2027.
For more information, contact Kathy Jedrziewski, jedrzmk@pennmedicine.upenn.edu
Download the full LOI request here.
Funded Projects
Principal Investigator: Roy H. Hamilton, MD, MS
Abstract: Communication deficits are present in the majority of persons with Alzheimer’s disease and are the hallmark of logopenic primary progressive aphasia (lvPPA), a condition typically associated with underlying Alzheimer’s disease pathology. While transcranial direct current stimulation (tDCS) holds promise as a treatment for language deficits in lvPPA, its progress toward widespread clinical use has been slow and inefficient. A major barrier to clinical translation of this safe, easy-to-use form of noninvasive brain stimulation is the complexity of its parameter space: stimulation characteristics like current intensity, polarity, and duration likely interact with disease-related factors like site and degree of neurodegeneration in complex ways. It is not currently feasible to systematically test these parameters because of the number of conditions that would need to be tested and the slow pace of clinical trials. Thus there is a critical need for computational strategies that can simulate the effects of different stimulation protocols on individuals’ brain networks—prior to patient experimentation—to optimize tDCS delivery. We hypothesize that data-driven virtual models can simulate persons with lvPPA, enabling accurate predictions of language performance both before and after neuromodulation. We propose a multidisciplinary pilot study that tests two distinct but integrable simulation approaches. For both analyses, we will leverage a large existing dataset from a recently completed R01-funded clinical trial of tDCS in PPA. The first approach will be to simulate brains with progressive loss of functional and structural integrity (“virtual network degeneration”) and the effects of tDCS on these brains by manipulating network variables in brain structural connectomes. Our second approach will be to simulate the impact of tDCS on “digital twins” of individuals with lvPPA that are constructed using multimodal data including clinical and laboratory data, linguistic and other psychometric performance measures, and MRI imaging, potentially including network data from our first approach. For both analyses, we predict that we will be able to use simulation to identify predictors of response to tDCS treatment.
This work will be conducted in close collaboration with two key colleagues: (1) Dr. John Medaglia (Drexel University), a close colleague who will apply his deep expertise in network analytical approaches and (2) Dr. Nishant Sinha (Penn Neurology), who will adapt his multimodal neurocomputational modeling pipelines developed for epilepsy surgery to neurodegenerative disease. If successful, this work will greatly advance tDCS treatment for lvPPA and will lay the groundwork for optimizing stimulation for more typical presentations of Alzheimer disease as well as other degenerative conditions affecting cognition. I look forward to hearing further from you regarding this outstanding opportunity.
Principal Investigator: James Shorter, MA, PhD
The overarching goal of this study is to develop a conditional gene therapy that selectively targets and reverses TDP-43 and tau aggregation in Alzheimer’s disease (AD) using a synthetic system that senses TDP-43 loss of function and drives expression of potent protein disaggregases—engineered Hsp104 or cytoplasm-localized PARLSkd3—only in neurons experiencing TDP-43 proteinopathy. This strategy directly targets TDP-43 proteinopathy, which is closely linked to accelerated cognitive decline in AD. By restoring TDP-43 function through conditional disaggregase expression, we aim to intervene at a critical mechanistic point in disease progression. The selective, pathology-responsive design of this system ensures precision, safety, and scalability for clinical translation.
Principal Investigators: Ian A. Blair, George Kannarkat, David J. Irwin, Katheryn A.Q. Cousins
Anti-amyloid β drugs have provided a breakthrough in Alzheimer’s disease (AD) therapy because they delay cognitive decline. However, they have limited influence on cognitive performance, which highlights a need to develop new therapeutic approaches to treat AD. Using cell models, we propose to lay the foundation for a novel treatment of AD via inhibition of ferroptosis and calreticulin (CRT) translocation by the nuclear exportin 1 (XPO1) inhibitor selinexor, and other inhibitors of high mobility group box 2 (HMGB2) activity.
HMGB2 is structurally related to HMGB1, the danger associated molecular pattern (DAMP), which has been associated with innate immunity, inflammation and neurodegeneration. HMGB1 and HMGB2 are the most abundant non-histone proteins found in the nucleus. We recently showed that secretion of HMGB2 protein during ferroptosis causes the translocation of CRT from the endoplasmic reticulum (ER) of cells to the plasma membrane (Fan J, Gillespie KP, Mesaros C, Blair IA. Commun Biol. 2024;7:1234). CRT translocation is a key process in the phagocytosis of neurons by microglia, which contributes to the pathology of AD. Recent studies have revealed that HMGB2 is secreted during ferroptosis of human SH-SY5Y neuronal and HMC3 microglial cells. In addition, we recently found that AD patients have highly elevated HMGB2 levels in CSF relative to healthy controls. We propose to confirm that HMGB2 protein is a viable drug target to restore neuron-glia dysfunction in AD, and to discover therapeutic approaches to inhibit HMGB2-mediated CRT translocation. This will set the groundwork for future studies to validate the role of HMGB2 in animal models of AD.
We anticipate that the proposed research will result in the discovery a therapeutic agent that (like selinexor) prevents HMGB2-mediated translocation of CRT from the ER to the plasma membrane of CNS cells. The therapeutic agent would inhibit microglial-induced phagocytosis, prevent neuron loss, and so provide a novel approach to stop the progression of AD.
Principal Investigators: Christopher Brown, MD, PhD, Dawn Mechanic-Hamilton, PhD, ABPP/CN, and David Wolk, MD
The goal of this study is to provide personalized monitoring of beneficial and adverse treatment response in individuals receiving anti-amyloid therapies for early Alzheimer’s disease. Investigators hope to discover which neuroimaging, blood-based, and cognitive measures best predict beneficial outcomes after treatment and which measures predict development of adverse outcomes.
Results of this study could be used to help guide decisions around treatment cessation and continuation, provide personalized safety monitoring that is less burdensome than current requirements, and identify mechanisms that may help enhance beneficial effects and/or reduce adverse side effects of anti-amyloid therapy.
“This is the first study to examine detailed imaging and cognitive measures longitudinally and explore individual differences in treatment response to anti-amyloid therapies,” said Christopher Brown, MD, instructor of Neurology at the University of Pennsylvania and principal investigator of the study. “Prior studies have been limited to looking at group effects that are not easy to translate to the individual patient level in the clinic.”
Principal Investigators: Zissimos Mourelatos, MD, Kelvin Luk, PhD, and Virginia M.-Y. Lee, PhD
The overarching goal of this project is to develop novel RNA therapeutics for the major human neurodegenerative diseases such as Alzheimer’s Disease (AD) and Related Dementias, Parkinson’s Disease (PD) and related Synucleinopathies.
Deposition of various combinations of pathogenic proteins is commonly seen in neurodegeneration. A promising therapeutic strategy is to reduce the levels of proteins that aggregate in neurodegenerative diseases by targeting their mRNAs.
“We developed a new platform for potent, in vivo gene silencing based on bitargeting linked small interfering RNAs (BlisRNAs) that can simultaneously silence two genes,” said principal investigator Zissimos Mourelatos, MD, Professor of Pathology and Laboratory Medicine. “BlisRNAs are simple and economical to synthesize and compatible with automated, large-scale production.”
So far, Dr. Mourelatos and his team have developed potent BlisRNAs that silence Microtubule-associated protein tau (MAPT), amyloid precursor protein (APP) and Synuclein Alpha (SNCA).
They are conducting biodistribution, safety, and efficacy studies of their BlisRNAs in relevant mouse models of AD and PD with the hope that this BlisRNAs will reduce pathology in a sustainable manner, without toxicity. “Since we developed each BlisRNA to be able to target all mRNA isoforms of human, mouse and primate ortholog genes, we can quickly test the same BlisRNAs in primates and ultimately in human clinical trials, if our studies in mice are successful,” explained Dr. Mourelatos.
Principal Investigators: Michael Haney, PhD, Saar Gill, MD, PhD, and Frederick “Chris” Bennett, MD
“Variants in the gene APOE have been shown to dramatically decrease the risk of developing Alzheimer’s disease (AD), however there are severe lipid related side effects outside the brain due to APOE expression in the peripheral immune system,” said Michael Haney, PhD, Assistant Professor of Pathology and Laboratory Medicine and principal investigator of this study. “The goal of this project is to develop engineered myeloid cell therapies that introduce AD-protective APOE variants into brain resident immune cells while sparing the peripheral immune system.”
Dr. Haney and his fellow investigators are examining whether replacing microglia with protective APOE variants is sufficient to inhibit progression of AD pathology in mouse models of AD -- and if so, which APOE variants are most effective in this.
Their work combines recently developed microglia replacement approaches with large scale CRISPR based gene editing of stem cells which enables them to quickly test the effect of many different variants of the APOE gene in AD progression. “This combination of this cell therapy approach with gene editing of the APOE gene is the first of its kind,” said Dr. Haney.
Principal Investigator: Xiaolu Yang, PhD, Professor of Cancer Biology, Perelman School of Medicine
The overarching goal of this project is to develop a gene therapy that restores protein homeostasis in Alzheimer’s disease (AD), addressing the underlying cause of this devastating disease.
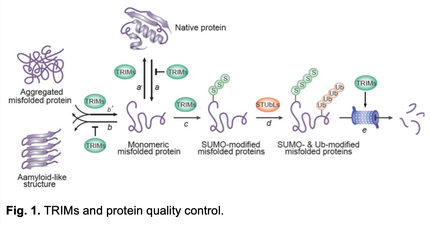
Description: AD is a disorder of protein misfolding and aggregation, characterized by extracellular plaques consisting of the amyloid b (Ab) peptide and intracellular neurofibrillary tangles (NFTs) consisting of tau protein. Our ability to develop effective therapies is impeded by limited knowledge of what drives protein aggregation in AD and how protein homeostasis can be therapeutically restored in patient brains. Research in my lab in recent years has revealed a metazoan-specific, major PQC system that is multifunctional, highly effective, and mechanistically distinct1-6. This system comprises members of the large family of tripartite motif (TRIM) proteins. We show that in an ATP-independent manner, individual TRIMs function as both molecular chaperones to prevent protein aggregation (Fig. 1, a, b) and disaggregases to dissolve pre-existing amyloid deposits (Fig. 1, a’, b’)5, 6, thereby maintaining solubility of normal proteins. TRIMs also promote proteasomal degradation of defective proteins through sequential SUMOylation and ubiquitination (Fig. 1, c, d)2, 5, 6 and directly activate the proteasome (Fig. 1, e)4, thus linking misfolded proteins with the proteasome. Together, these TRIM activities provide several safeguards that may synergize to buffer the toxicity of misfolded proteins.
The client proteins for the TRIM system are broad, including tau6, a-synuclein5, polyQ proteins2, and TDP- 43 (our unpublished results). Among TRIMs, TRIM11 possesses especially strong PQC activities4-6. Of note, TRIM11 is markedly downregulated in AD brains, and its expression is inversely correlated with NFT burden6, suggesting that TRIM11 downregulation contributes to AD pathogenesis6.
CNS diseases have been recalcitrant to conventional pharmacological treatments. In recent years, gene therapy has become an important approach, and its potential is emphasized by positive clinical outcomes. The multiple and potent PQC activities endowed in individual TRIMs, along with their downregulation in neurodegenerative diseases, make them especially attractive as transgenes to restore protein homeostasis, thus addressing the root cause of these diseases. Indeed, we showed that intracranial delivery of the TRIM11 gene via AAV ameliorated pathology, neuroinflammation, and cognitive impairments in multiple animal models of tauopathies and synucleinopathies5, 6. These findings provide a proof of concept for a gene therapy for AD.
Nevertheless, the AAV-TRIM11 vector has only been performed in rodents5, 6. Rodent brains are very different in size, structure, and AAV transduction characteristics from brains of large animals including humans. To translate this gene therapy from the bench to the bedside, we propose to test AAV-TRIM11 vectors in the cat, which is a well-established model for neurological studies, has a gyrencephalic brain structure like human, and is more readily available than non-human primates.
We will test different AAV-TRIM11 vectors in the cat brain for the transduction efficiency and toxicology. Specifically, we will test different serotypes including AAV9 (which is commonly used for CNS tissues) and AAV1 (which shows a better transduction efficiency in some brain regions). To avoid potential toxicity to dorsal root ganglion (DRG), we also will test a vector that incorporates the target sequence of miRNA183. As miRNA183 is expressed specifically in the DRG, the incorporation of such target sequence would suppress the expression of the transgene in this tissue. This vector will be compared to the vector without the miRNA183 target sequence. Once an optimal AAV vector is selected, we will test different doses and perform pathology and expression analysis, to identify a dose with maximal gene transduction and minimal toxicity.
Potential to result in or enhance AD therapeutics: Upon the completion of the proposed experiments, we are ready to perform IND-enabling, GLP study for this novel gene therapy approach.